Written on: February 1, 2018 by W. Stephen Tait
Hello, everyone. Spray packages allow consumers to easily dispense a controlled amount of product to a wide range of areas. Corrosion is an issue for all types of spray packaging and packaging materials.
However, corrosion can be controlled and/or prevented with a comprehensive corrosion prevention and control program. Let’s start with a brief overview of why spray package corrosion occurs and then discuss the elements in a comprehensive corrosion control and prevention program.
Three elements are needed for spray package corrosion:
Corrosion is the degradation of materials and all materials are susceptible to some form of corrosion. In other words, all spray packages can corrode and there’s no such thing as a corrosion-proof material. Corrosion often causes materials to lose important properties, such as the barrier property of polymer coatings.
The environment inside spray packages consists of a formula. Water is one of the most common ingredients in spray formulas, but is corrosive toward all materials and may be present as a contaminant as well as part of the formula.
Water molecules readily absorb on polymer surfaces and diffuse into polymer materials, subsequently causing polymer corrosion such as blistering. Water is also electrochemically active and removes electrons from the surface atoms of uncoated metals and metals under coatings and laminated films.
All spray packages have internal surfaces exposed to the formula; surface molecules are thermodynamically unstable and thus susceptible to corrosion. Corrosive formula ingredients adsorb on surfaces, absorb into the polymers, diffuse through polymers and remove electrons from both uncoated and coated surface metal atoms (i.e., corrosion). Material corrosion initiates and propagates after one or more of these steps or with all four steps in series.
A comprehensive corrosion control and prevention program has six elements as shown in Figure 1. Our Elements of Spray Package Corrosion short course provides a detailed discussion of this Figure, so only an overview will be provided here.
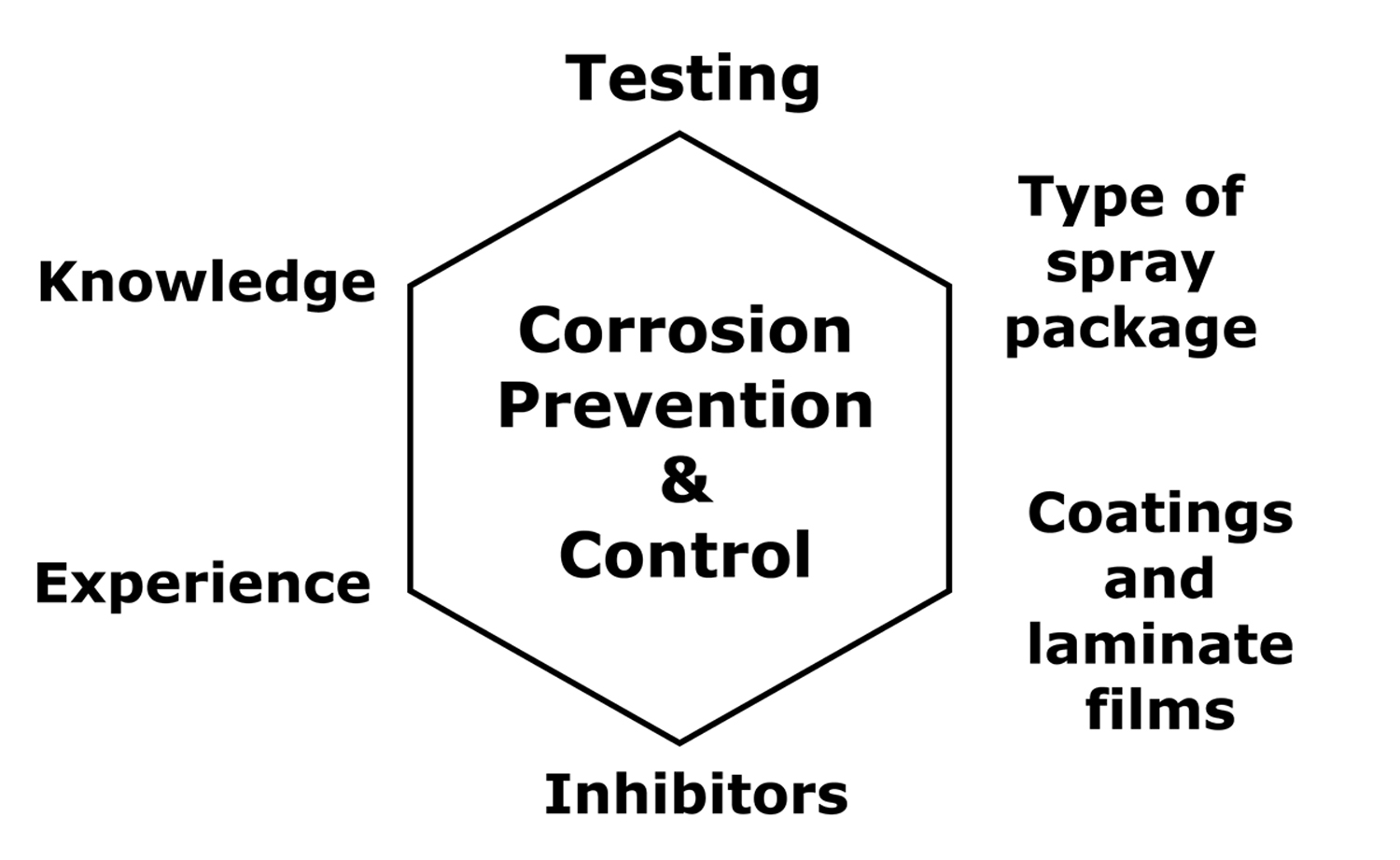
Figure 1: The elements of a comprehensive spray packaging corrosion control
and prevention program.
Corrosion Testing
The current state-of-the-art for corrosion science is extensive, but not enough to predict if corrosion will or will not occur using data tables, equations or first principles. Consequently, corrosion testing is an essential element in a corrosion control and prevention program. Corrosion tests could be either a storage stability test, or an electrochemical corrosion test.
A storage stability test has an approximately 93% correlation between its results and actual commercial spray package corrosion when the test is conducted for at least one year. Lower test times reduce the correlation and thus increase the risk of surprise corrosion with commercial units. In addition, corrosion rates are not accelerated by exposing packages to higher temperatures. In other words, results from higher temperatures cannot be used to predict package service lifetimes.
Electrochemical corrosion tests are completed in less than 100 days with a correlation that exceeds 99% when the appropriate protocols and test cells are used. In other words, electrochemical tests are completed in less time than a storage stability test with less risk. Electrochemical tests can be applied to all types of spray packages and spray package materials.
Types of spray packages
There is a variety of spray package types available for spray formulas. Sometimes one type of package is significantly more corrosion resistant than others.
Corrosion testing is used to determine if new prototype and derivative formulas are corrosive toward specific package types, select the most corrosion resistant form of spray packaging and determine if alternate packages are suitable for current formulas.
Internal coatings & laminate films
Uncoated metals are the least expensive form of spray package materials. However, in some instances an internal coating either prevents or extends the service life of a metal package to an acceptable level. Corrosion testing selects the most corrosion-resistant form of coating or laminate film for a spray package-formula combination.
Corrosion inhibitors
Corrosive formulas do not have to be abandoned. Corrosion inhibitors are an effective way to control or prevent spray package corrosion, although their inhibitor development often extends a development timetable. However, corrosion inhibited formulas are often patentable and thus provide a market advantage. Corrosion testing is used to select the most effective corrosion inhibitor and determine its effective concentration range.
Experience & Knowledge
Knowledge without practical experience or experience without theoretical frameworks are incomplete and will not result in useful, predictive professional practice. In other words, experience and knowledge are intricately linked.
Extensive corrosion databases for spray packaging are unavailable in the public domain because of market competition. Consequently, proprietary corrosion databases are typically developed by individual companies only for internal use. A proprietary database allows a) faster-to-market introduction of new and derivative products; b) provides formulation guidelines for avoiding developing corrosive formulas; c) reduces corrosion risk during product and derivative development; and d) reduces the corrosion risk of commercial products.
Pair O Docs has a state-of-the art electrochemical corrosion testing laboratory; please contact me if you would like to know more about our rapid, predictive corrosion testing. You can also visit our new website which has a short Vision Video that discusses all our corrosion prevention and control services. Please also contact me if you would like to a have our Elements of Spray Package Corrosion short course taught at your R&D facility. Back issues of Corrosion Corner are available on CD-Rom. Thanks for reading and I’ll see you in March. SPRAY