Written on: July 1, 2021 by W. Stephen Tait
Hello, everyone. Last month, I began a series on material defects in spray packaging and the relationship between these defects and corrosion. We completed a discussion on the more common defects in aluminum aerosol containers. This month, I will discuss the more common material defects found in laminated foil bags and tinplated steel aerosol containers.
Laminated aluminum foil bags
Figure 1 has an example of a micro-bulge in the laminate film on the aluminum foil. This type of material defect is common with the laminated aluminum foils used to fabricate the internal laminated bags for aerosol containers. The range of technical difficulties associated with cross-sectioning this type of defect has prevented us from more thoroughly examining it. Consequently, we currently have not been able to determine the properties of –and the most likely causes of—film micro-bulges. It has been our experience that micro-bulges do not contribute to or cause laminated aluminum foil bag corrosion.

Figure 1:, Bulge in the laminate film on aluminum foil.
Figure 2 provides an example of a polymer film that delaminated at the bag weld. It has been our experience that this type of material defect is rare and can be avoided with optimized welding. We have not observed instances where this type of material defect contributes to or causes spray package bag corrosion. However, this type of defect could cause bag rupture at the delaminated area.
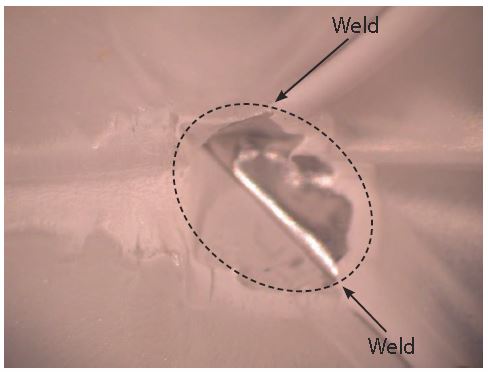
Figure 2: Delamination of laminate film at bag welds.
Figure 3 provides an example of a crack in the aluminum foil under the laminate film. In our experience, this type of material defect is typically associated with a single manufacturing batch of laminated foil bags. We have not observed instances where this type of material defect contributes to or causes laminate foil bag corrosion.
It is possible that a bag with this type of defect might leak product. However, the product would have to diffuse through both the inner and outer laminate film layers in order for the bag to leak. We have not observed instances of this type of leaking at this time.

Figure 3: Crack in the aluminum foil used for internal spray package laminated film bags (the crack is illuminated from behind).
Tinplated steel aerosol containers
Figure 4 provides an example of an area where the tin coating did not cover (wet) the steel substrate. Non-wetting produces holes in the tin coating that expose either the substrate steel or a very thin iron-tin alloy layer. Numerous holes in tin coatings, such as that in Figure 4, are present in all tinplated steel aerosol containers.
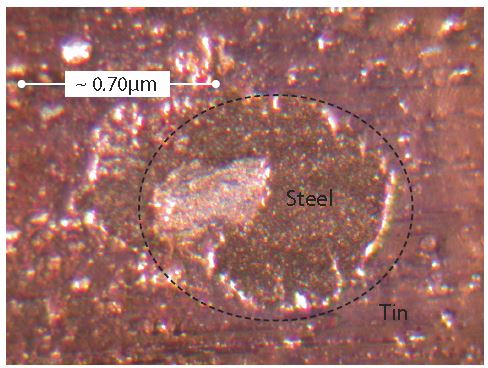
Figure 4: Steel not wetted (covered) by tin.
There are two different morphologies for the holes in tin coatings. Figure 4 shows the traditional type of hole morphology and Figure 5 shows a newer type of hole morphology that has appeared within the last two decades.
Notice when comparing Figure 4 and Figure 5 that the newer holes are more symmetrical than the traditional holes. Most likely the newer, symmetrical holes arise from either a new composition of tinplating bath chemistry or a new tinplating process.
Holes in tin coatings are potential sites for pitting corrosion. However, the chemical composition of a formula determines if holes in tin coatings will or will not result in pitting corrosion.

Figure 5: Newer type of hole in the tin coating for tinplated steel (Note: not all holes are noted with arrows).
It is unknown at this time if the traditional hole morphology or the newer hole morphology results in different susceptibilities to and/or magnitudes of pitting corrosion. Corrosion testing is the only way to determine if a given formula will cause pitting corrosion at holes in the tin coating.
Steel aerosol container welds are diffusion welds formed with heat and pressure. The heat is produced from an electrical current flowing between the overlapping ends of the tinplated steel sheet used to form the cylindrical container body. In some instances, the combination of pressure and heat is not optimum and a small amount of metal is ejected from under the overlapping ends.
Figure 6 has an example of a small particle of metal ejected from a weld. This phenomenon is referred to as weld spatter. Weld spatter is not common. However, pitting corrosion could occur if weld spatter produces a small cavern in the weld. The chemical composition of a formula determines if pitting corrosion will occur in a cavern formed by weld splatter.

Figure 6: Weld splatter.
The examples in this column and in SPRAY’s June 2021 Corrosion Corner both contain examples of macro-defects that can be seen either with the unaided eye or a light microscope. Next month, I will complete this series with discussions of micro-defects that cannot be seen with the unaided eye and their relationship to spray package corrosion.
Thanks for your interest and I’ll see you in August. Please contact me at 608-831-2076; rustdr@pairodocspro.com; or from one of our two websites: pairodocspro.com and aristartec.com. SPRAY